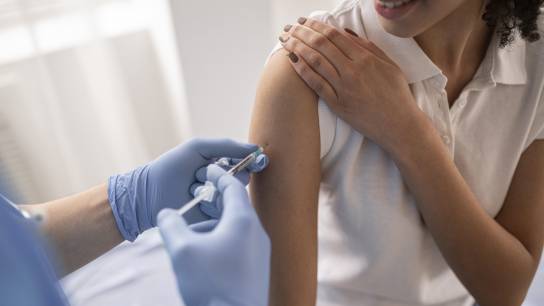
First Patients Enrolled: 3TR Asthma Biologics Cohort Aims to Identify Novel Immunological Treatment Targets in Severe Asthma
The 3TR Asthma Biologics Cohort (ABC study) is a pan-European prospective translational study aiming to improve personalized medicine for patients with severe asthma. By investigating possible immunological mechanisms underlying non-response or incomplete response to treatment with current biologics, the ABC study team aims to find new therapeutic approaches to help those affected by severe asthma. With the first four enrolled patients from Denmark and one additional targeted participant, the study has now successfully started.
The Asthma Biologics Cohort is part of the COPD and Asthma disease cluster in 3TR (Work Package 8) and is jointly led and coordinated by three institutions:
- Bispebjerg Hospital (BBH) – Prof. Celeste Porsbjerg
- Academic Medical Center Amsterdam (AMC) – Prof. Anke-Hilse Maitland
- European Respiratory Society (ERS)
In the ABC study, a real-life cohort of a total of 300 severe asthma patients from asthma centres across Europe will be monitored for three years. At the beginning, each of the patients receives one of the currently approved biologics for the treatment of severe asthma for the first time, with the treatment continuing constantly over the study period and its effect on the patient's disease being comprehensively assessed and accompanied by biosampling. Regular examinations of the patients serve to determine three types of response to the prescribed asthma biologics: super-responders, clinical responders and non-responders. With the accompanying sampling, possible biomarkers/predictors for the successful or unsuccessful administration of the drug can be found. The latter can then serve as the basis for a targeted improved medication (in the case of clinical responders) or for completely new treatment approaches (in the case of non-responders).
Eight research institutions from six countries are already collaborating on the 3TR Asthma Biologics Cohort and more are expected to join by the end of 2022:
- Denmark (Bispebjerg Hospital)
- Netherlands (Academisch Ziekenhuis Groningen, Academic Medical Center Amsterdam)
- UK (University of Leicester, University of Glasgow)
- Poland (The Medical University of Lodz)
- Belgium (Ghent University Hospital)
- Sweden (Karolinska Institutet)
In addition to acquiring further participating research institutions by the end of this year, the team around Prof. Porsbjerg and Prof. Maitland wants to reach the target of 300 patients for the cohort by the end of 2023 and have completed the first comprehensive examination of the patients after one year of medication by the end of 2024.